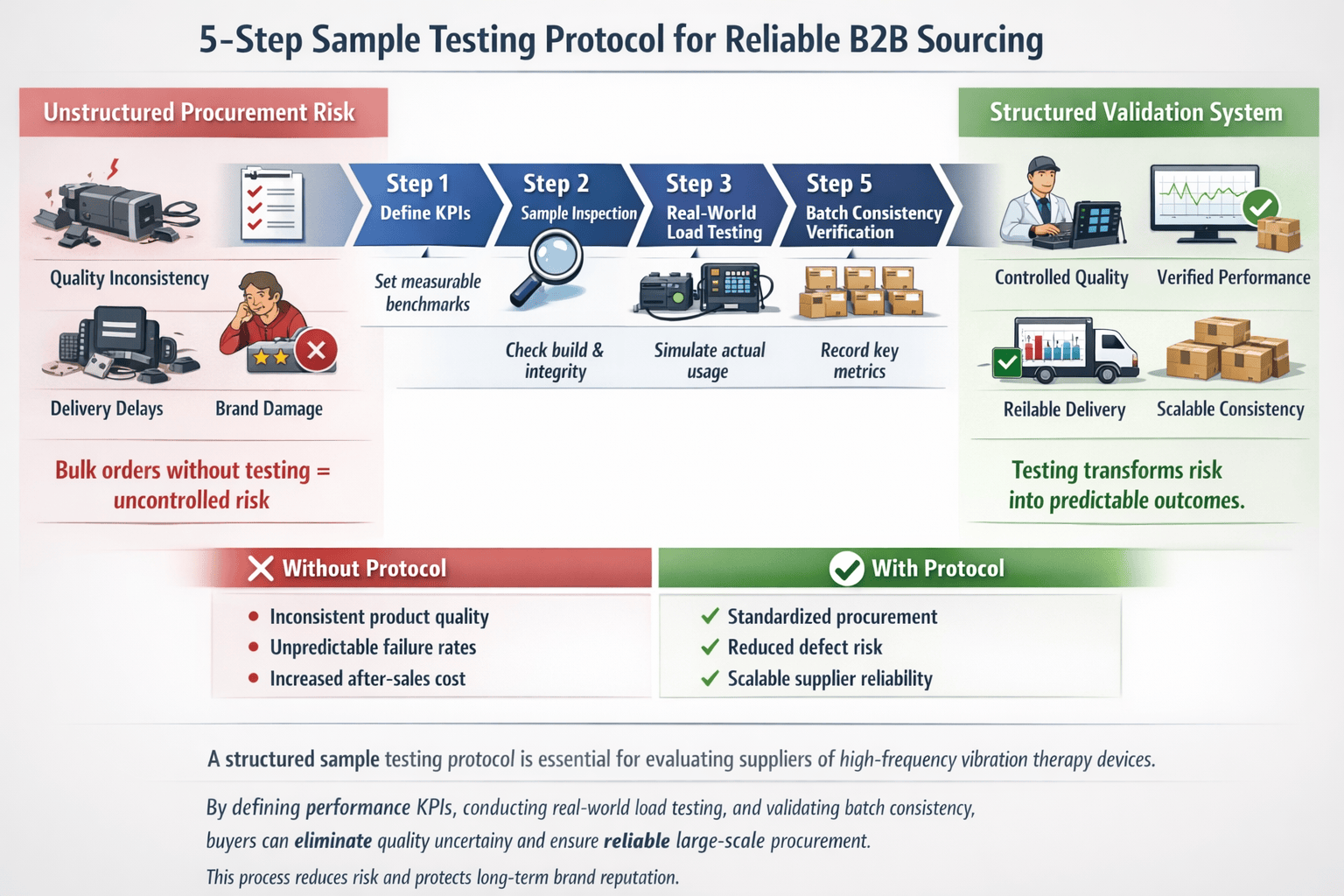
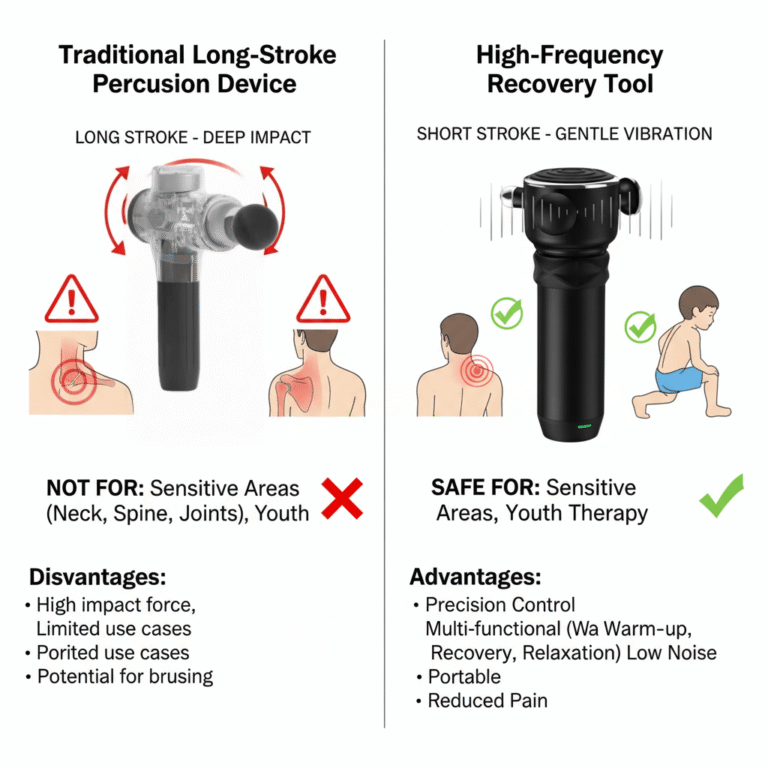
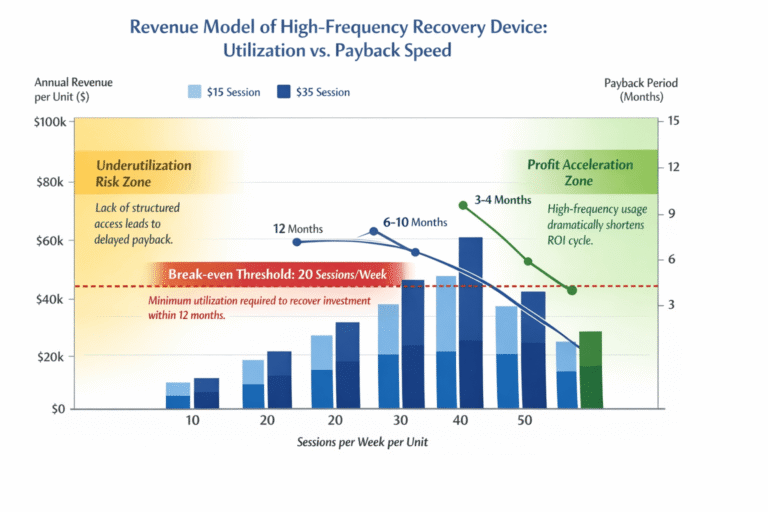
When evaluating a new supplier for high-frequency 3D vibration therapy devices1, a rushed bulk order2 can expose your business to quality inconsistencies3, performance issues, and delivery delays. Without a structured sample test4, you risk substandard products that damage brand reputation and customer trust. A validated testing protocol ensures that your partner delivers consistent, reliable performance from sample to mass production.
The proven 5-step sample test4ing protocol begins with defining key performance indicators5, proceeds through real-world load testing, and ends with data-driven decision-making. Each step ensures alignment between sample and final batch output, focusing on frequency stability, vibration consistency, and long-term operational reliability—all critical for B2B sourcing decisions.
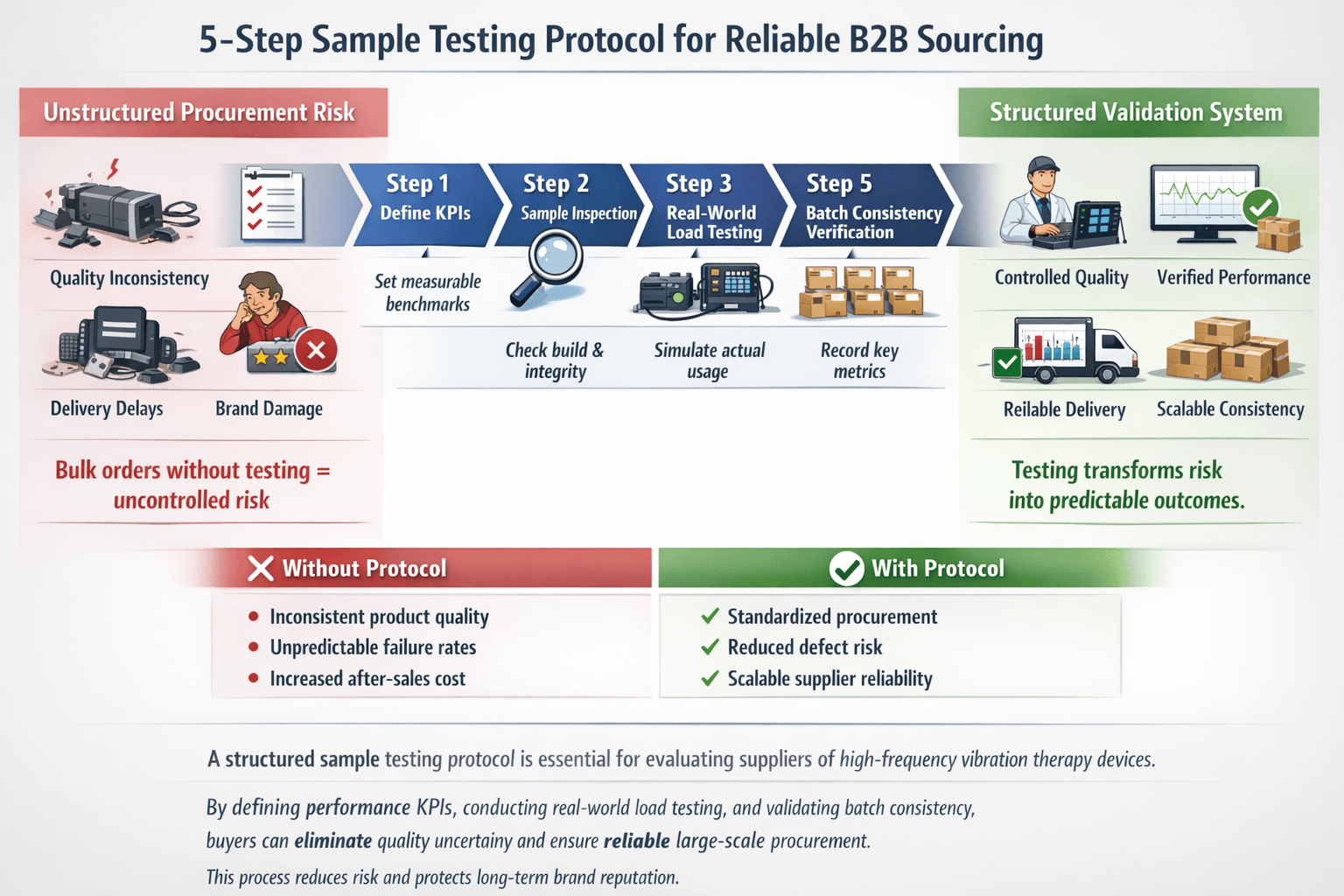
This structured approach is essential for procurement teams sourcing medical-grade wellness equipment. By testing actual performance under controlled conditions, you reduce sourcing risk and validate supplier capability6. The following steps guide you through engineering-level verification7—ensuring your bulk order2 meets expectations from the first shipment.
What to Measure First: Key Performance Indicators for High-Frequency Output and Consistency?
You must test measurable technical parameters before evaluating user experience. Relying on subjective impressions risks accepting variation that scales into batch nonconformance. A clear, objective benchmark is essential for technical due diligence.
The first measurable indicators are vibration frequency range8 (RPM), amplitude stability9, and output consistency10 across all five speed levels. These must be recorded using a vibration analyzer11 or oscilloscope12. Frequency must remain within manufacturer-rated specs under no load and light load, with minimal deviation.
A systematic evaluation begins when you receive the first sample. I always check the following immediately:
- Frequency accuracy at lowest (4500 RPM), midpoint (5500 RPM), and highest (7000 RPM) settings.
- Amplitude consistency across all five gear (settings), particularly focusing on low-amplitude modes used for sensitive areas.
- Vibration waveform stability with no drift, jitter, or unintended harmonic spikes.
These metrics directly reflect the quality of the motor, drivetrain, and control firmware. Any deviation suggests under-spec components or unstable power supply design—both common issues in low-cost manufacturing.
| Parameter | Acceptable Range | Test Method | Supplier Deviation Threshold |
|---|---|---|---|
| Frequency (RPM) | ±50 RPM of rated value | Oscilloscope + signal analyzer | >100 RPM deviation |
| Amplitude (mm) | ±0.2mm nominal | Laser displacement sensor | >0.3mm variation |
| Duty Cycle Stability | ≤5% fluctuation | Electronic load testing | >10% ripple |
| Starting Response Time | <1.2 sec | Timer + power-on trigger | >2 sec delay |
I’ve seen suppliers who claimed 7000 RPM output fail to exceed 6700 RPM under light load—due to poor spindle bearing tolerance and underpowered motor drives. Nutritional devices aren’t the only category where real-world performance reveals off-spec parts.
Integrating these measurements early prevents costly rework later. Vibration output must be repeatable and within datasheet claims before evaluating comfort or accessory fit.
Simulating Real-World Use: Evaluating Performance Under Load and Continuous Operation
A steady test bench reading means little if the device fails during prolonged use. Real-world operation involves thermal load13, mechanical stress14, and variable torque demands—conditions that expose design flaws missed in idle testing.
High-frequency 3D vibration therapy units must sustain performance through continuous 10-minute operation at medium-high settings (5500–7000 RPM). During this test, monitor temperature rise at the motor housing, power draw variation, and vibration consistency. A stable unit shows minimal frequency drift (<3%) and no abrupt noise changes.
In one real case, a supplier passed initial RPM tests but failed under sustained load. The motor overheated in 4.2 minutes, triggering thermal shutdown. The root cause? An insufficiently rated heat sink and low-efficiency motor winding.
The following engineering parameters are critical under load:
- Thermal rise: Temperature should not exceed 55°C at the motor housing after 10 minutes of continuous operation15. Hot spots >60°C indicate inadequate cooling or thermal interface material.
- Power draw stability: A constant 12–16W draw across five levels suggests consistent driver efficiency. Sudden spikes suggest inefficient current regulation.
- Mechanical resonance: Listen for chatter or harmonic noise increase. These signals mechanical looseness in the spindle or mass imbalance.
- Battery voltage drop: Measure voltage under load. A drop >1V from nominal (5V) indicates poor internal chemistry or BMS inefficiency.
I use a test rig with fixed load torque simulators to mimic muscle resistance. The setup includes a precision force sensor and feedback loop that maintains consistent kinetic resistance across all runs.
| Test Condition | Target | Observer Metric |
|---|---|---|
| 10-min continuous run | 7000 RPM at 3rd gear | Frequency variance ≤3% |
| 5-min operation at 4th gear | 6000 RPM | Temperature ≤55°C |
| Load step-up from 1→5 gear | 200ms response | No delay or stall |
| 30-sec rapid cycling | 4→3→5→2→4 | 100% cycle completion |
These conditions replicate actual use in fitness centers and clinical settings. A device that fails here will suffer field failures—especially in high-usage environments.
From Sample to Decision: How to Interpret Test Results for Confident Bulk Purchasing
Failure in the test phase must be traced back to root cause—and assessed for risk. Did the deviation stem from a bad sample, a flawed design, or a production inconsistency?
A successful sample must meet all pre-agreed technical specifications and demonstrate consistency across three identical test units. If variation exceeds 3% in key parameters, even across samples, reject the batch. Consistency is non-negotiable for bulk order2s.
I’ve seen multiple quotes rejected not because of one failure, but because the supplier couldn’t explain why temperature rose above 60°C in two out of three samples. The root cause? Inconsistent application of thermal paste during assembly.
This leads to three decision rules:
- Immediate rejection if any unit exceeds thermal or frequency thresholds.
- Conditional acceptance only if supplier provides a root-cause analysis and corrected sample within 48 hours.
- Full approval only after 3 consecutive samples pass all tests with ≤1% deviation.
Before placing a bulk order2, I verify:
- Are test logs signed and dated?
- Was the test performed by a neutral lab, or was it a "soft validation" in-house?
- Are all parameters auditable with time-stamped data?
If the supplier lacks traceable data, I’ll delay the order—even if the sample "feels okay." Without digital proof of compliance, there’s no way to enforce quality in mass production.
Always compare the sample’s test sheet against the engineering spec sheet. Discrepancies in amplitude tolerance, frequency sweep curve, or power draw indicate divergent design or sourcing.
The cost of correcting a failed batch is typically 8–12× the cost of initial testing.
Conclusion
A disciplined sample test4 protocol separates reliable suppliers from uncertain ones. By measuring high-frequency output, stress-testing performance under load, and validating consistency across iterations, you build confidence in bulk order2s. This process isn’t optional—it’s foundational to engineering-grade sourcing.
-
Explore this link to understand the technology and benefits of high-frequency 3D vibration therapy devices. ↩
-
Explore the potential risks associated with bulk orders and how to mitigate them. ↩ ↩ ↩ ↩ ↩
-
Explore the causes and implications of quality inconsistencies in manufacturing processes. ↩
-
Learn about the importance of sample testing in ensuring product quality and supplier reliability. ↩ ↩ ↩
-
Discover how key performance indicators can guide your product evaluation process effectively. ↩
-
Understand the criteria for evaluating supplier capability to ensure quality products. ↩
-
Learn about the importance of engineering-level verification for ensuring product quality. ↩
-
Find detailed methods for measuring vibration frequency range to ensure product compliance. ↩
-
Understand the significance of amplitude stability in ensuring consistent product performance. ↩
-
Explore techniques to maintain output consistency for reliable device performance. ↩
-
Learn about vibration analyzers and their role in assessing device performance. ↩
-
Discover how oscilloscopes are used in testing to measure electrical signals accurately. ↩
-
Explore the concept of thermal load and its impact on device performance during testing. ↩
-
Understand the effects of mechanical stress on devices and how to mitigate risks. ↩
-
Learn about the importance of continuous operation testing for product reliability. ↩